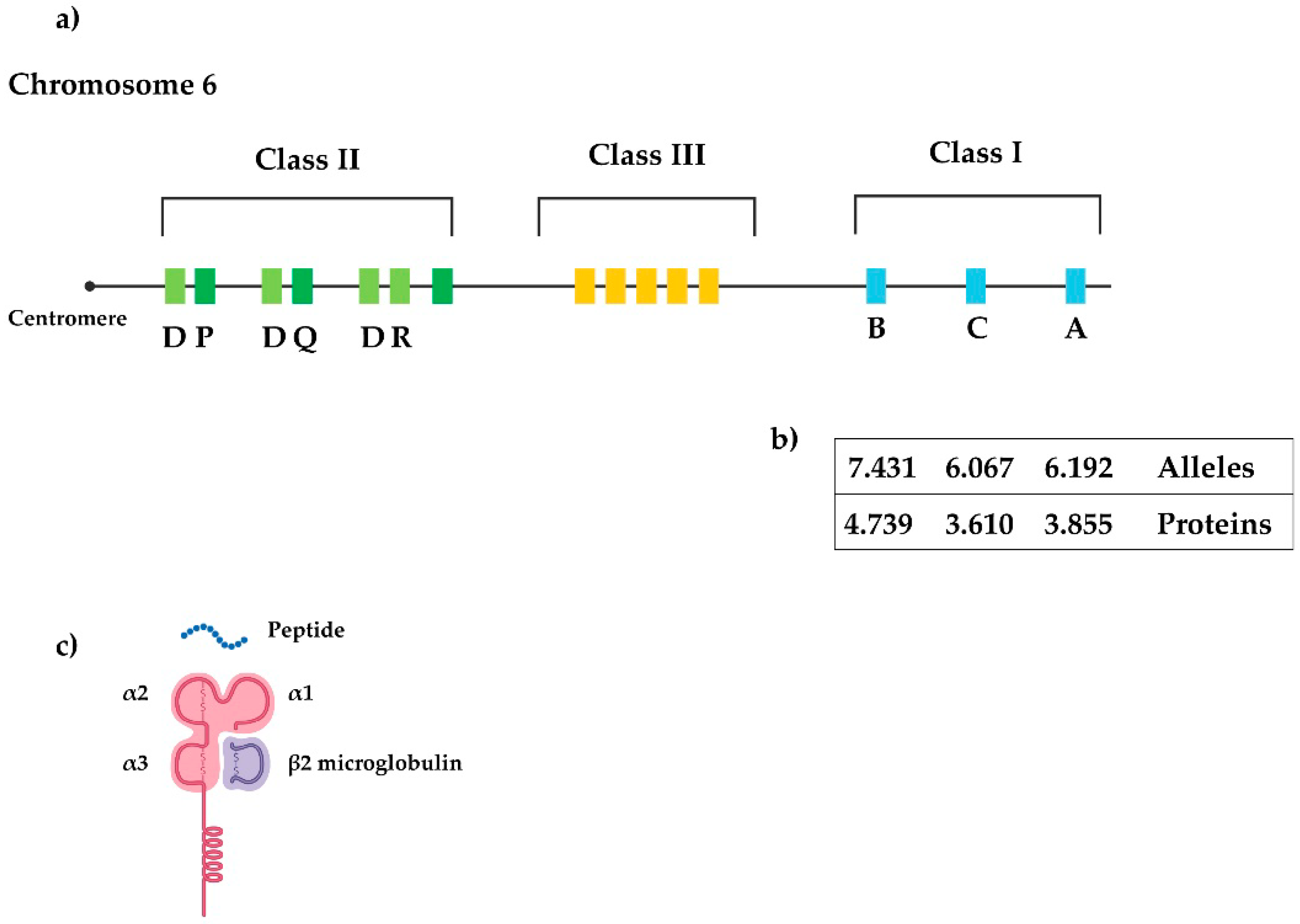
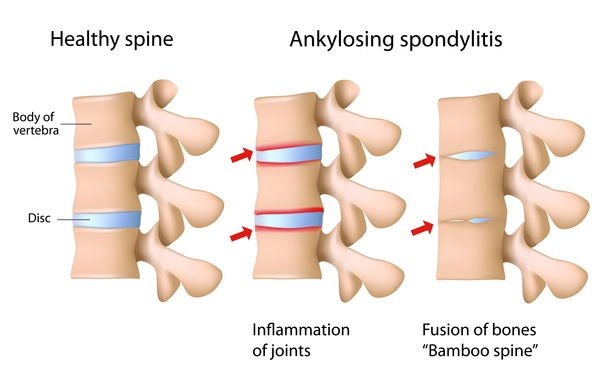
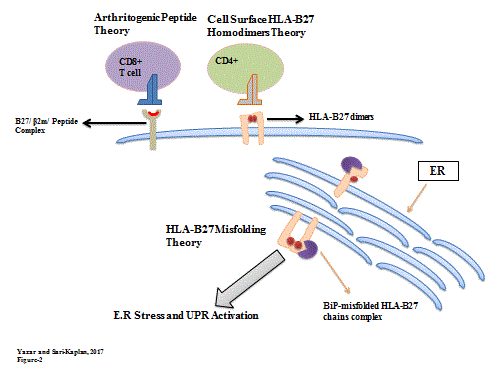
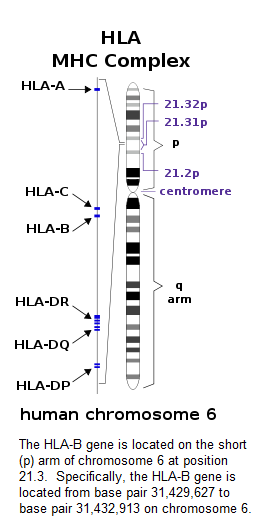
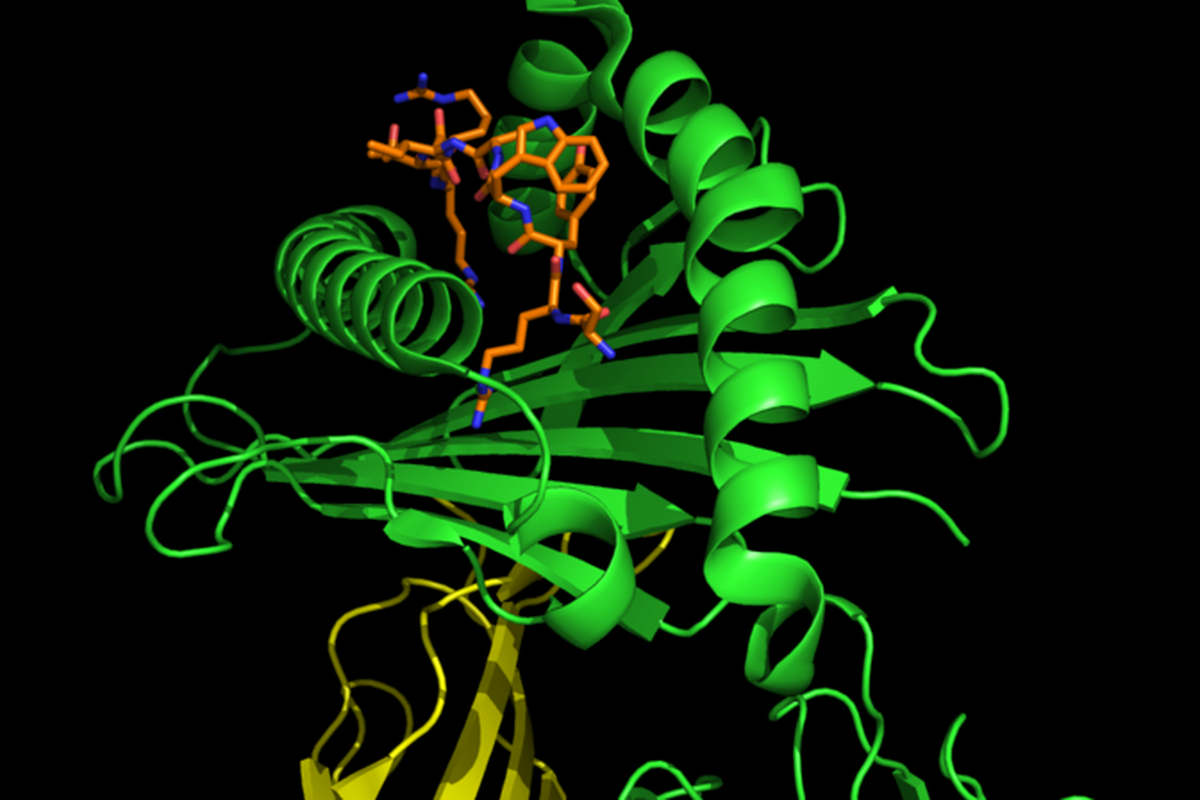
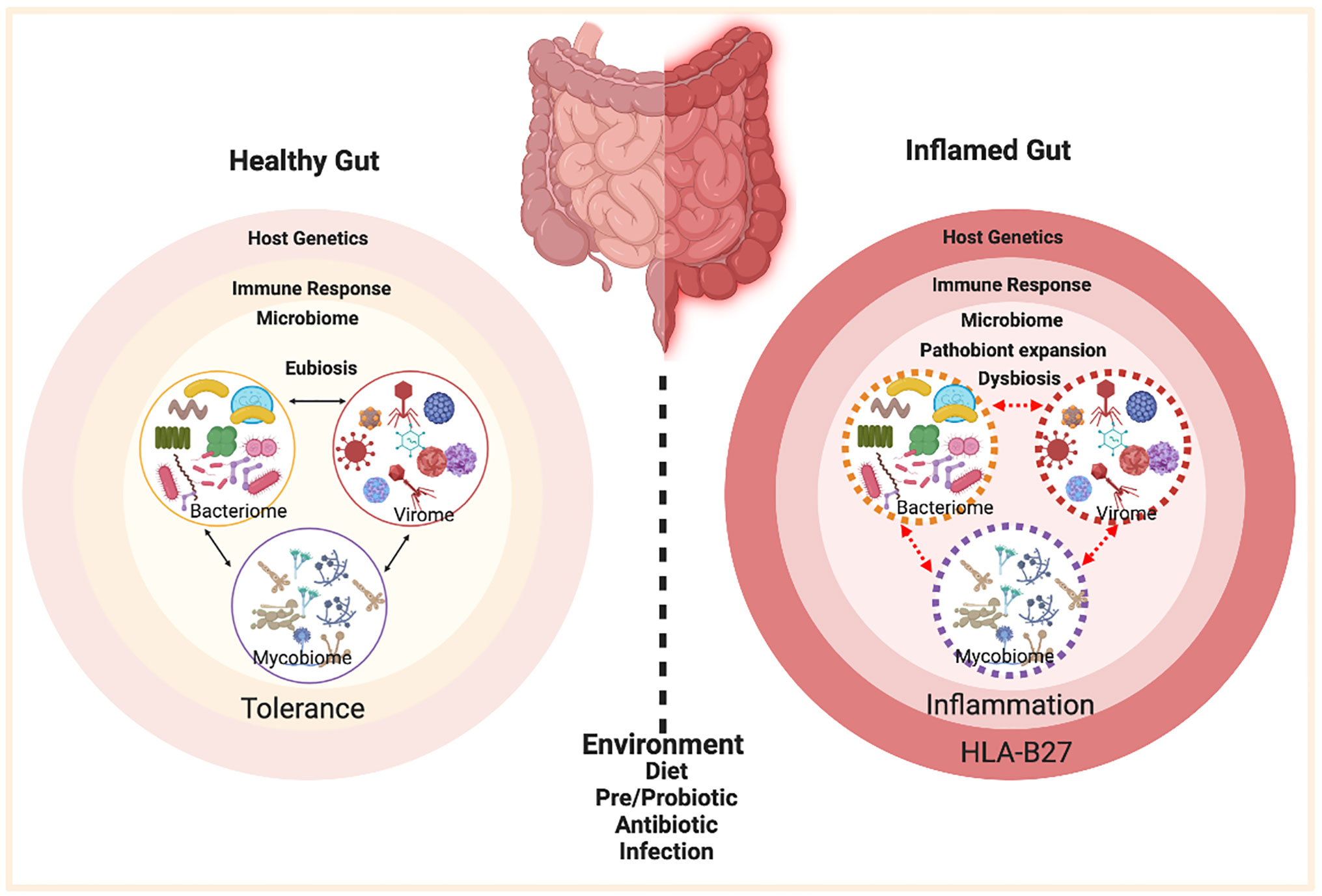
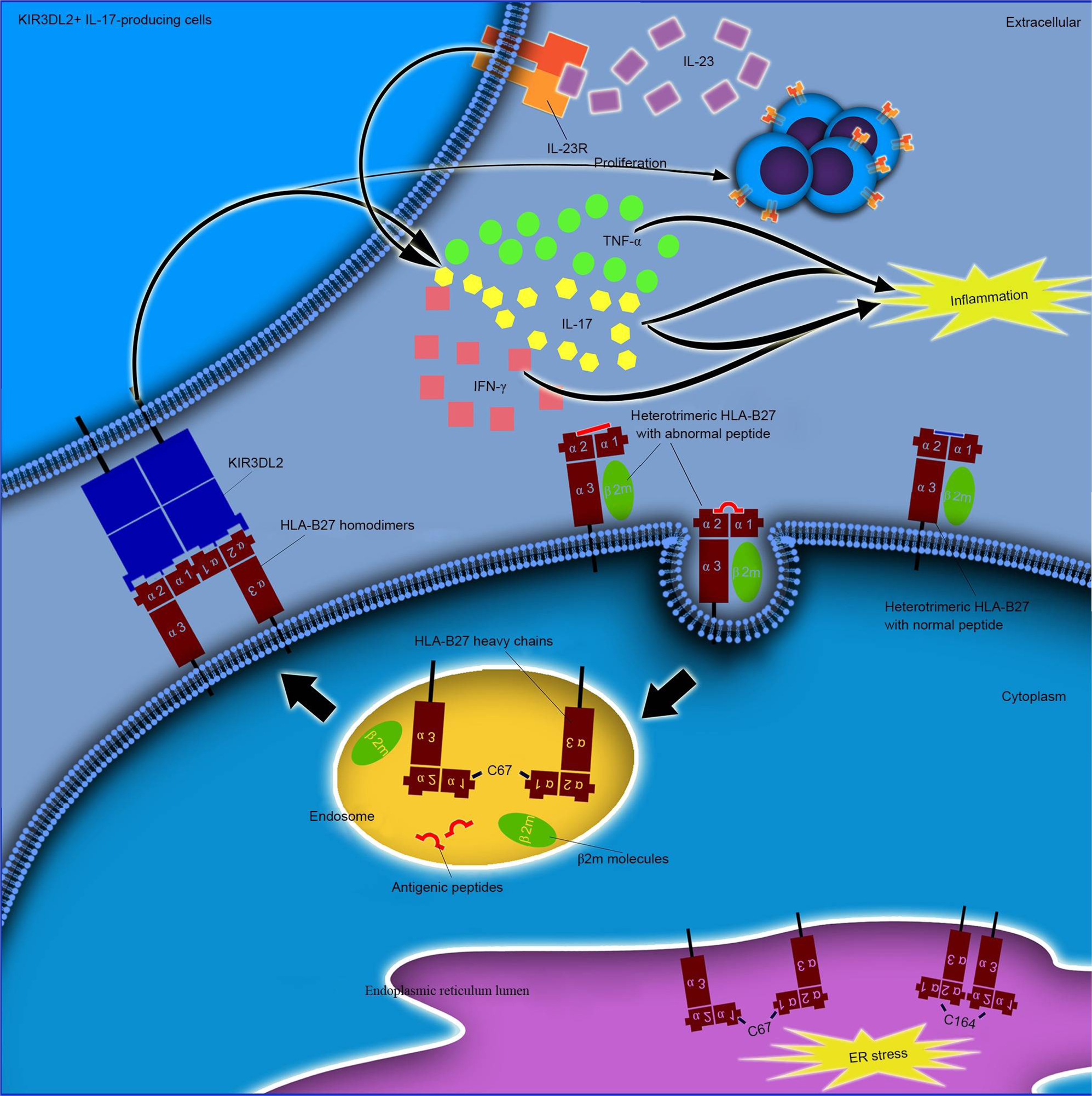
The interplay between HLA‐B27 and ERAP1/ERAP2 aminopeptidases: from anti‐viral protection to spondyloarthritis - Vitulano - 2017 - Clinical & Experimental Immunology - Wiley Online Library

The role of HLA-B27 polymorphism and molecular mimicry in spondylarthropathy: Molecular Medicine Today

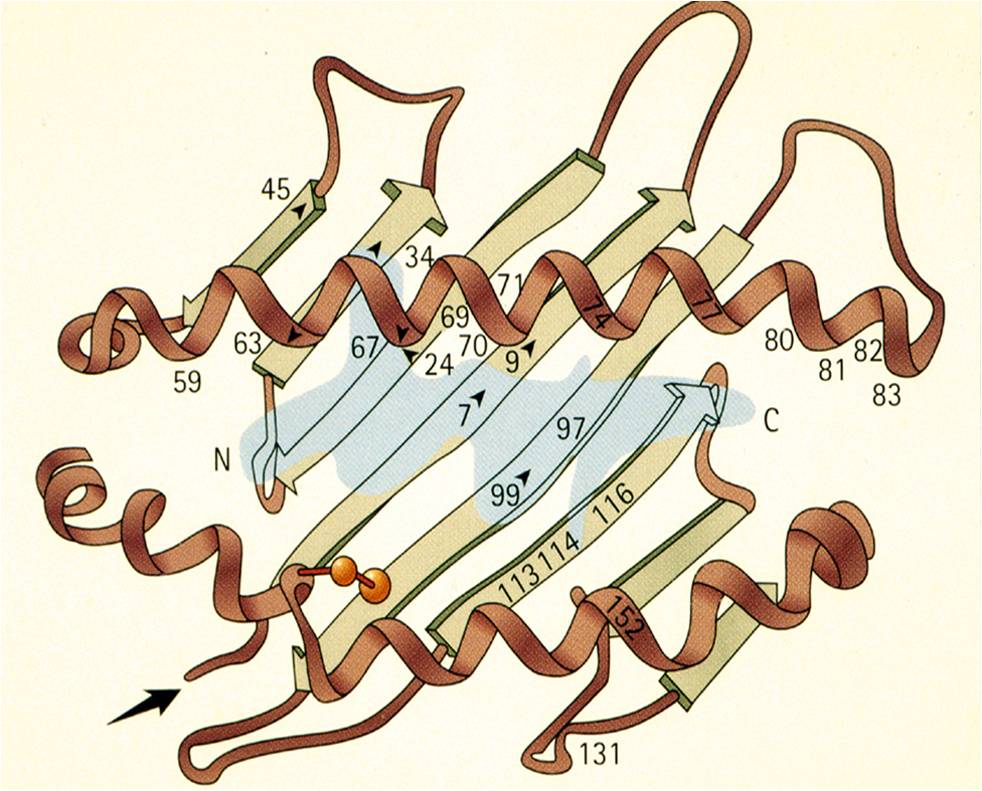
RCSB PDB - 1HSA: THE THREE-DIMENSIONAL STRUCTURE OF HLA-B27 AT 2.1 ANGSTROMS RESOLUTION SUGGESTS A GENERAL MECHANISM FOR TIGHT PEPTIDE BINDING TO MHC