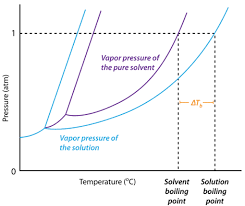
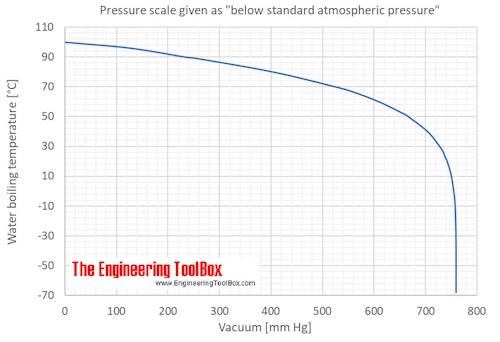
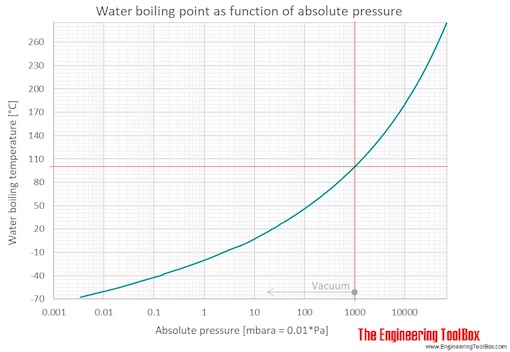
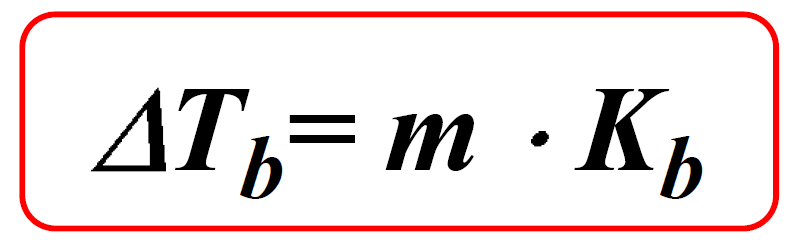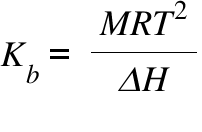Calculate the freezing point and the boiling point at 1 atmosphere of a solution containing 30 g cane sugar (molecular mass 342 ) and 150 g water.Given : Kb = 0.513 and Kf = 1.86

Boiling Point Elevation and Freezing Point depression - Example 2 ( Video ) | Chemistry | CK-12 Foundation
GitHub - stuartmcelhany/boiling-point-calculator-peng-robinson: Peng-Robinson Boiling Point Calculator